What is the equivalent mass fraction of a 5M saline solution?
A 5M molarity NaCl solution is equivalent to a salt-water solution prepared at mass fraction of 24.53 wt%.
Is 5M NaCl more than saturated saline solution?
I did not understand how anyone could make a 5M (molarity) NaCl solution, like this one: UltraPure™ 5M NaCl. It seemed to me that this was beyond saturation concentration of 26 wt% at room temperature. But I didn’t know how to confirm my suspicion. And the web was mum on this issue.
5M NaCl stock solution is a common concentration used in labs to produce exact dilutions. the concentration is expressed in weight of solute to volume of the total solution (not solvent). Molarity (M) is frequently used in lab situations. It is expressed in the unit of g/L (gram/liter).
On the other hand, when mixing salt in everyday situations, mass fraction wt% is more often used. For instance, 23.3 wt% salt water provides the lowest freezing point at -6°F. A saturated saline solution at room temperature has a mass fraction of 26 wt%. Seawater for a marine aquarium is prepared with a 3.5 wt% solution. Mass fraction wt% is denoted as a pure percentage number with a unit of mass-to-mass ratio (w/w).
I just wanted to know the wt% of a 5M NaCl solution, so I can figure out whether 5M NaCl is less concentrated than, exactly equal to, or impossibly more concentrated than a saturated saline solution (26 wt%).
How much water does it take to make 1 litter of 5M NaCl?
NaCl has a molecular weight of 58.44g. So 5M = 58.44g * 5 = 292g.
A 5M NaCl solution in 1 liter (1,000 mL) means 292 g salt solute in a 1,000 mL solution (not solvent). Now, I don’t know the amount of solvent (water) this solution needs. It’s not a straightforward subtraction, because chemical reactions are funny. And 1,000 is in milliliter, while 292 is in gram. But I do know that the solvent amount is not 1,000 mL, from my recent experience making saline solutions of different concentrations. I do regret not having carefully measured the volume of resulting solutions before. Else I would have had an empirical answer to this.
Mass fraction operates in a different space. With mass fraction, the amount of solvent is known by weight. In theory, saturated saline solution at room temperature is 26 wt% (percentage by weight). To make a saturated saline solution, one mixes roughly 350 g salt with 1,000 g (aka 1,000 mL) water. And 350 / (350 + 1000) = 26 wt%. The resulting amount of solution is between 1,000 mL and 1,350 mL.
Now, for my 5M NaCl question, we have a situation where the resulting solution has a volume of 1,000 mL. But I do not know how to back out the volume of solvent needed. It is not 1,000 mL – 292 g. If it were, then 292 g salt to 708 g (or mL) water solvent clearly far exceeds the saturation level, as 292 / (292 + 708) = 29.2 wt%.
But what “is” the actual solvent amount needed, such that 292 g of salt with the solvent produces 1,000 mL of 5M solution? It seemed to me that I would need to figure out the solvent (water) amount, in order to calculate the equivalent wt%.
What am I missing?
I realize that wt% and M are fundamentally incompatible. wt% operates in weight space for solute, solvent and solution. Thus the equation “solute weight + solvent weight = solution weight” works well with wt%.
We cannot apply this equation to M because it operates in weight/volume space. And the same equation does not hold. That is, “solute volume + solvent volume != solution volume”. Thus I cannot convert the concentration of 5M to wt% space.
Density of 5M solution is the missing piece
Finally it dawned on me that I had overlooked the fact that the 1,000 mL solution in 5M NaCl did not weigh 1,000g. It is not water anymore. It is not true that one can’t back out the volume of the solvent in molar equations, even if chemical reactions are funny and multivariate. If I assume a particular room temperature, atmosphere pressure, etc., then I only need one extra piece of information to bridge the molar volume space and the wt% weight space: the density of the 5M solution.
If I have the density, then I can back out the weight of the 5M solution. Since I will then be working in the weight space, I can simply derive the ratio wt%.
Putting the puzzle together
Now that I know that I am looking for density, I can find the density of 5M NaCl at a few places, including this product properties page.
| Concentration | 5M |
| Density | 1.190 g/cm3 |
One liter of solution at 1.190 g/cm3 density is 1,190 g. I don’t need to bother with the solvent after all. I can now calculate mass fraction wt% directly:
292 g / 1,190 g = 24.53 wt%
However, if I insisted on knowing exactly how much water solvent is used, I could back it out easily:
1,190 g solution – 292 g salt = 898 g water
Since 898 g water occupies 898 mL at room temperature, this gives us how much water is needed without using a scale.
Wolfram Alpha
I found no help on Google, before I figured this out myself. I still can’t find any discussion about this problem on Google, even after I figured this out, and know what to search for.
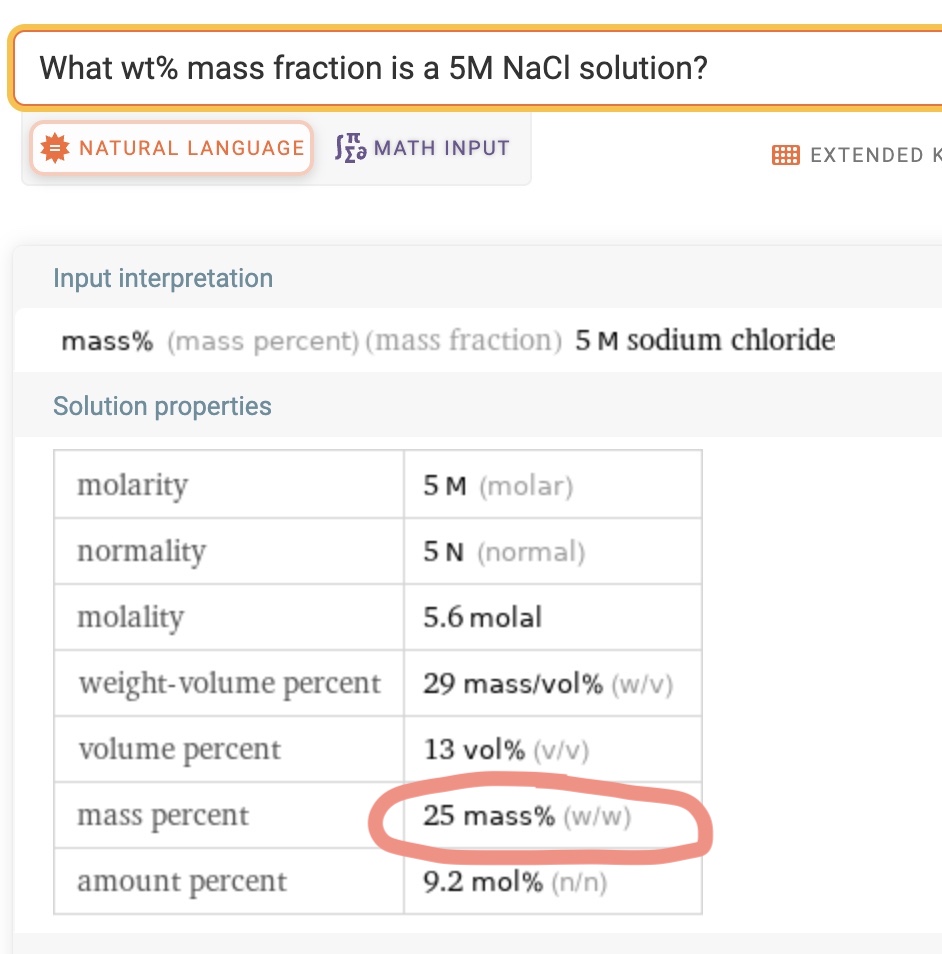
However, I typed “What wt% mass fraction is a 5M NaCl solution?” into Wolfram Alpha just now, after having written all of above.
It shows a 25 wt% as mass faction for a 5M solution. How does it calculate this? If it uses density of the 5M solution, where does it get that from?
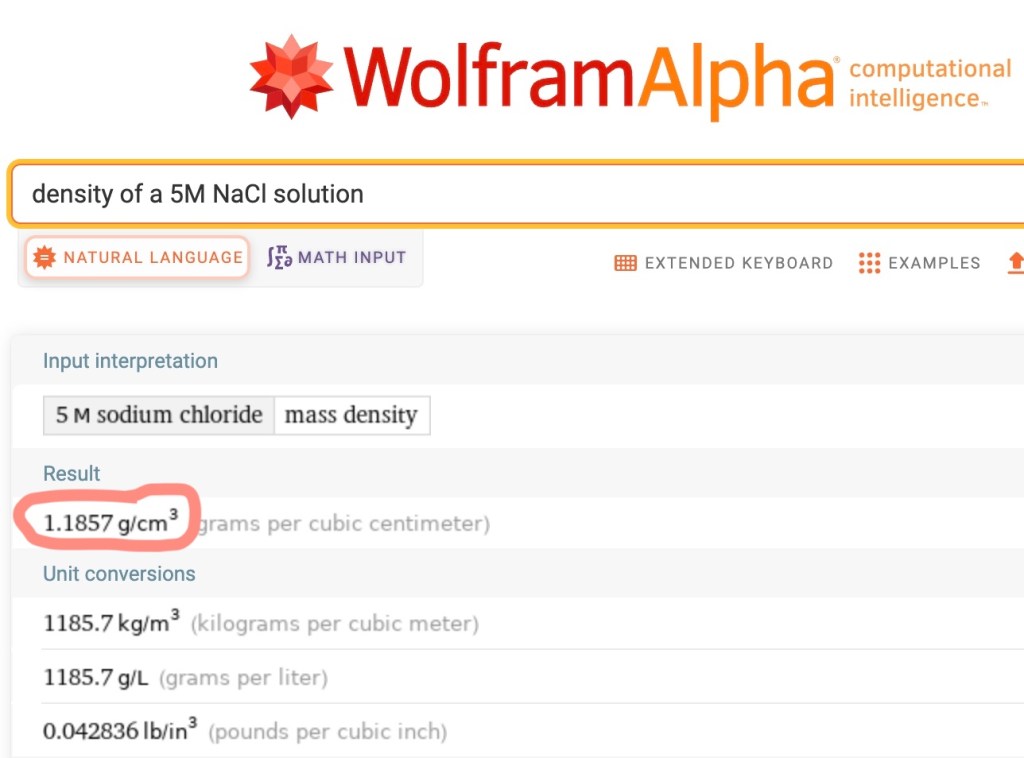
It turns out Wolfram Alpha does know about the density of a 5M NaCl solution, as shown on this query page for “density of a 5M NaCl solution”. It says the density of 5M NaCl is 1.1857 g/cm3 which seems right.
But it still doesn’t answer the question, “how does it derive this density?”
Partial molar property and volume
I alluded to funny chemical reactions between solutes and a solvent earlier. This is known as a partial molar property. Various terms such as partial molar volume and apparent molar volume describe the observed, non-linear change in solution volume. I suspect that the “density of 5M solution” shown by Wolfram Alpha is derived from these curves, somehow. But I am yet to find a source that defines these curves at room temperature under a normal environment.
Engineering ToolBox
u/SOwED on Reddit pointed out the excellent graph and conversion page at Engineering ToolBox on Densities of Aqueous Solutions of Inorganic Sodium Salts. This one page bridges mass fraction (wt%), molality (mol/kg), and molarity (mol/liter), with clear graphs and tables, using density as the connection between them. It shows these graphs and tables for many salts, including sodium chloride. I only wish it had more data points than 5, 10, 15 and 20 wt% for NaCl. With that being said, it is clearly possible to interpolate points in between those shown in tables, judging by the almost-linear curves shown in these plots.
ChatGPT
Ten months after the initial writing of this article, ChatGPT becomes a thing (Jan 2023). After chatting with it and asking it a few different questions from topics I’ve written about on this blog, it quickly become obvious that ChatGPT is not a good tool for answering questions. It is best used to create fictional narratives at this stage. It is too good at natural language, and too assertive in its narratives that one can never know what parts of its output are made up. It never divulges sources, thus one can never confirm anything it dreams up. In a way, it operates like an average human being.
I asked ChatGPT: What is the equivalent mass fraction of a 5M saline solution?
It just made up some bullshit and confidently proclaimed: